|
Seoul-based Seegene has grown into one of the world¡¯s leading developers of multiplex molecular diagnostics (MDx) products, exporting to more than 37 global markets including the U.S.A.
¡°Our respiratory virus diagnostics has been approved by 30 countries (including Canada and EU) as an IVD (in vitro diagnostics); it is currently in the process of acquiring America¡¯s FDA approval along with other major products, a sign that the FDA approval will enable Seegene to penetrate the North American market (which comprises over 50% of total molecular diagnostics market) in acceleration,¡± said Chun Jongyoon,
the President of Seegene Inc.
Seegene¡¯s advanced MDx technologies overcome the limitations of conventional PCR and Real-time PCR. Seegene¡¯s DPO¢â‚ (Dual Priming Oligonucleotide), READ¢â‚ (Real Amplicon Detection) and TOCE¢â‚ result in MDx tests that detect dozens of different pathogens simultaneously with high sensitivity and specificity.
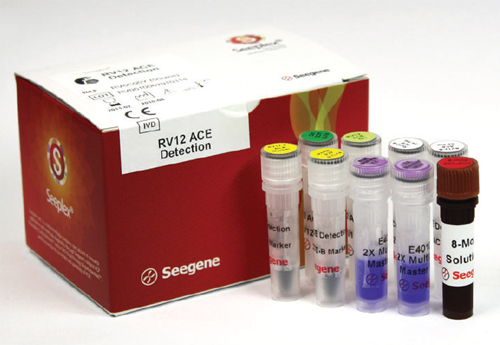
The company¡¯s flagship diagnostic product called ¡°Seeplex RV ACE Detection¡± employs PCR to amplify nucleic acids of target pathogens for maximized sensitivity and rapidly detects 12 different respiratory pathogens simultaneously. Clear and accurate results promote the most appropriate treatments while reducing unnecessary antibiotic administrations and hospitalizations.
Seeplex RV ACE Detection may be also used to identify and isolate infected patients to prevent nosocomial infection spread. ¡°Our diagnostic products are supplied to 70 domestic medical centers including university hospitals and 330 medical institutions and government organizations in 50 different countries¡±, said the President, adding that Seegene monopolistically
supplies the product to ~70% of the university hospitals in Korea. The number of domestic institutions using its product is expected to be very high.
Seeplex RV Detection can be used for usual surveillance of national respiratory virus and currently CDCs in 23 provinces in China and some of the stategovernmental testing centers operate year-round surveillance systems with its product.
¡°Seegene has been consistently striving to develop the leading-edge molecular diagnostic technology and products since its foundation in 2000¡±, he said, adding that Seegene now has got its own molecular diagnostic products that detect the pathogens causing infectious diseases such as Respiratory virus, TB/MDR, A/B/C Hepatitis, STI, Sepsis, Cervical Cancer, Meningitis, acute Gastrointestinal, H1N1 and somatic mutations including drug-resistant bacteria, Thyroid cancer, Leukemia and genetic mutations.
The leader added, ¡°Seegene has formed a strategic partnership with automatic instrument-specialized companies for the convenient result analysis and test process, strengthening its recognition in the world¡¯s markets via various business partnerships with global multinational companies.¡±
Until now, Seegene has won a total of 15 awards from government branches and civil evaluation organizations such as Jang Young Shil Award (the Ministry of Education, Science and Technology), New Growth Engine grand prize (the Ministry of Knowledge Economy), Healthcare Industry Technology prize (the Ministry of Health and Welfare), and Hidden Champion company (KRX).
Lastly, Seegene has been selected as one of the World¡¯s 30 Molecular Diagnostic Companies by various global medical market research institutes (Kalorama Information, Research and Markets, etc.).
|