|
On the cutting edge of medical science today is gene and stem cell
therapy, using an individual¡¯s own cells to combat a wide variety of
disorders. FCB-Pharmicell Co. has developed a cell therapy solution utilizing
somatic adult stem cells for treating acute myocardial infarction, spinal cord
injury and ischemic stroke.
Current treatment for acute myocardial infarction is limited to the
administration of drugs after the occurrence, and some patients have no
recourse other than a heart transplant or the use of an artificial heart for the
remainder of their lives. To overcome the limitations of current treatment
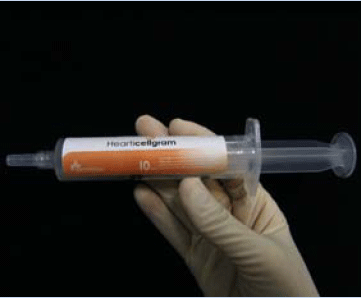
methods, FCB-Pharmicell¡¯s Hearticellgram-AMI was developed to harvest
the patient¡¯s own Mesenchymal stem cells (MSC), which are then cultivated and injected in the patient¡¯s coronary
artery. This treatment accelerates the formation of capillary vessels in the afflicted region and regenerates cardiac
muscle cells. Hearticellgram-AMI was cleared for phase II/III clinical trials for commercialization by the Korea
Food and Drug Administration in 2006, with trials taking place in leading Korean hospitals, including the
Severance Hospital, the Wonju Christian Hospital, and the Inha University Hospital.
|