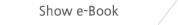|
CU Medical systems, Inc. specializes in the design, development, manufacturing and selling of high-tech emergency medical devices. Its core items include automated external defibrillators. Its latest semiautomated external defibrillator iPAD (Intelligent Public Access Defibrillator) has gotten approval from the U.S. FDA.
The i-PAD has been developed with focus on ease of use, simple operation and reliable systems so that anyone without professional training can save the life of people with Ventricular Fibrillation (VF) and Fast Ventricular Tachycardia (FVT). i-PAD provides simple and direct voice indications with an LED indicator for a straightforward rescue operation. Also, the device is designed to test itself on a regular basis - daily, weekly and monthly - so as to be ready for rescue job anytime and anywhere.
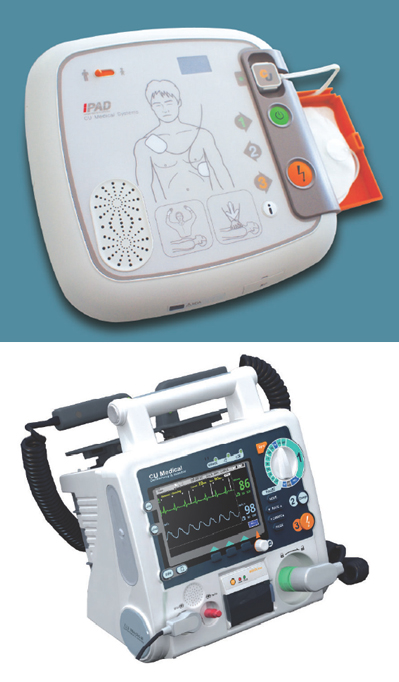
A brand-new CUHD1 has been released for medical professionals, including medical specialists, or paramedics to treat patients with a sudden cardiac arrest. Focusing on defibrillation and monitoring of the patient, the device has such features as the manual and automated external defibrillator (AED) operation, defibrillation using paddles or pads, synchronized cardioversion, Nellcor SpO2 pulse oximetry with alarm, ECG monitoring (3 Lead ECG/ 5Lead ECG /10 Lead ECG) and non-invasive pacing mode.
|